Lack of Key Transport Molecules May Lead to Nerve Fiber Loss in Neurodegenerative Diseases, Study Says
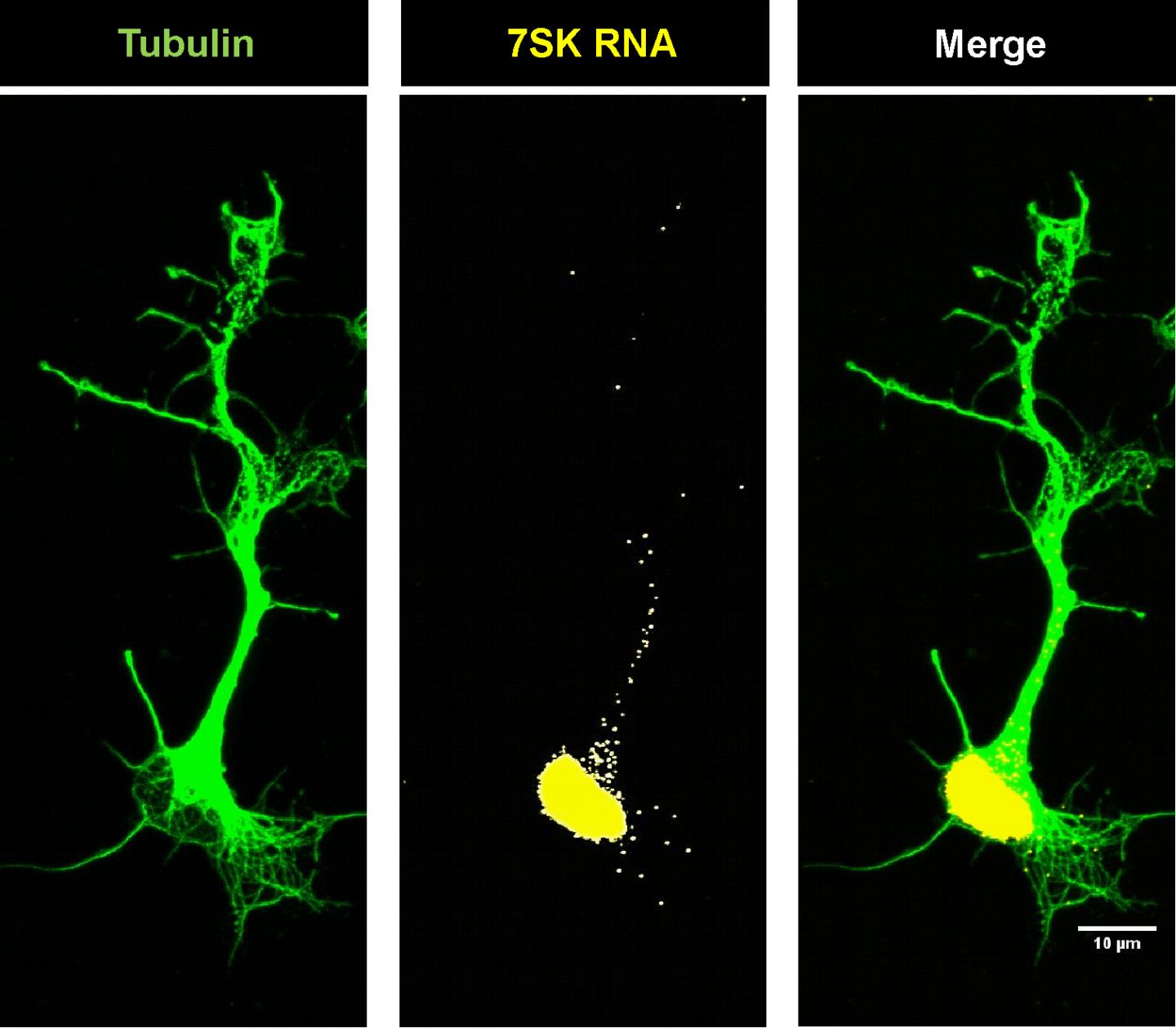
This is the localization of the ZSK-RNA in motor neurons whose shape was represented by tubulin, a structural protein. Credit: Hanaa Ghanawi
A disruption between two molecules that transport substances to the remote ends of special nerve fibers may result in neurodegenerative disorders such as spinal muscular atrophy (SMA) or amyotrophic lateral sclerosis (ALS), a study suggests.
The study, “hnRNP R and its main interactor, the noncoding RNA 7SK, coregulate the axonal transcriptome of motoneurons,” appeared in the journal Proceedings of the National Academy of Sciences (PNAS).
Motor neurons are essential to carry nerve impulses from the brain to the spinal cord and muscles. Their damage leads to the development of progressive disorders characterized by muscle atrophy.
“We know from previous studies that signal transmission defects from motor neurons to the muscle play a key role in disorders such as amyotrophic lateral sclerosis and [SMA],” Michael Briese, the study’s lead author and a scientist at the University of Würzburg’s Institute of Clinical Neurobiology, said in a press release.
These transmission defects are caused by impaired transport of molecules called messenger RNAs (mRNAs) in nerve fibers, or axons. These molecules are generated from the DNA in gene expression, the process that leads to the formation of new proteins. Their absence leads to a lack of proteins necessary for nerve endings to work properly.
“When the mRNAs are missing, the proteins can no longer be synthesized in this cell compartment which is crucial for the function of the nerve cells,” Briese said.
University of Würzburg researchers showed that a noncoding RNA molecule called 7SK is essential to this process. Noncoding RNAs are molecules whose genetic information is not translated into proteins.
Using novel cell culture systems, the investigators demonstrated that 7SK, along with hnRNP R, an RNA-binding protein, participates in the transport of important mRNAs. If one of the two molecules is missing, mRNA transport is disrupted, leading to less growth of nerve fibers and a defective link with muscles.
“These results propose a role for 7SK as an essential interactor of hnRNP R to regulate its function in axon maintenance,” the researchers wrote in the study.
“This may enable further improvements of new therapies which are already being used successfully in the hospital to treat [SMA] by creating options that may influence the composition and regulation of such RNA transport complexes in axons and motor neurons,” Briese said.
Besides the implications in SMA treatment, scientists also hope that more research will focus on the effect of disrupted mRNA transport in neurodegenerative diseases.
Briese and his team are now planning to study the extent to which 7SK and hnRNP R are altered in motor neuron disorders.







