Cure SMA Releases Updates On SMA Drug Discovery Programs

Cure SMA, a non-profit organization focused on supporting research projects to find a cure for spinal muscular atrophy (SMA), has released an update on its drug pipeline of products currently under development to treat SMA. The organization has revealed its enthusiasm with both the increased number of projects and the advancements accomplished.
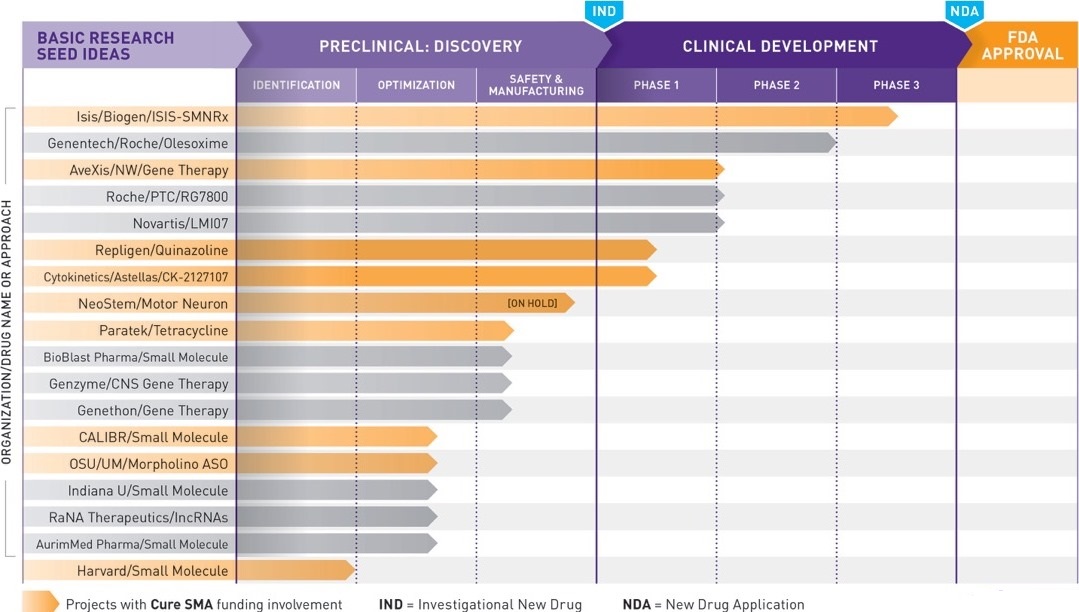
According to the updated data released by Cure SMA, the organization currently holds 18 active programs as well as seven programs in clinical trials and 15 partnerships established with pharmaceutical companies. There are a total of 26 programs on its cumulative pipeline, making it the highest amount of research projects and collaborations in the history of Cure SMA, a 15 year old organization.
In addition to the increased number of projects, Cure SMA is now responsible for financially supporting half of the ongoing programs developing new drugs to target SMA. “The current pipeline reflects the success of our research model. As more and more of the drug programs we’ve already funded have had success, we’ve attracted more and more investment from pharmaceutical companies,” explained the organization.
Cure SMA has attracted pharmaceutical companies to establish partnerships as a consequence of its programs’ potential, thus receiving seed funding. The organization also announced several companies already expressed their interest in initiating SMA drug programs.
So far, Cure SMA has already invested $57 million in SMA research, from which more than $25 million took place in the past 10 years. Despite the encouraging results, Cure SMA remembered that only 10% of the drugs studied in clinical trials are in fact approved by the U.S. Food and Drug Administration (FDA). Therefore, the organization is determined to find both funding and insurance coverage to support research and SMA patients.