Biogen Idec and Isis Pharmaceuticals Announce NURTURE, a Phase 2 Clinical Study for Patients With Genetic SMA
Written by |

 Biogen Indec and Isis Pharmaceuticals Inc. recently announced they will start recruiting patients for NURTURE, a multi-center, Phase 2 clinical trial that will evaluate the efficacy of ISIS-SMNRx (ISIS 396443) in pre-symptomatic newborns that have a genetic diagnosis of Spinal Muscular Atrophy (SMA).
Biogen Indec and Isis Pharmaceuticals Inc. recently announced they will start recruiting patients for NURTURE, a multi-center, Phase 2 clinical trial that will evaluate the efficacy of ISIS-SMNRx (ISIS 396443) in pre-symptomatic newborns that have a genetic diagnosis of Spinal Muscular Atrophy (SMA).
The primary objective of the study is to assess the efficacy of multiple doses of ISIS SMNRx (ISIS 396443) in preventing or delaying the need for respiratory intervention or death in infants with genetically diagnosed and presymptomatic SMA. Secondary objectives of this study include assessment of the clinical effects of ISIS SMNRx (ISIS 396443) in infants with genetically diagnosed and presymptomatic SMA.
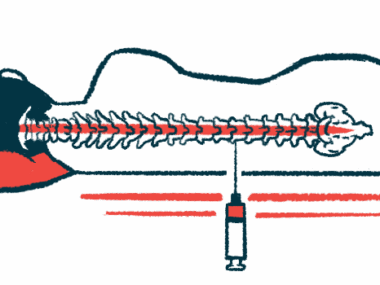
A total of 25 infants will be enrolled from centers around the world until April 2020. These include clinical centers in United States, Middle East, South America Europe, and Asia Pacific. All participants will receive the investigational drug as an intrathecal injection.
[adrotate group=”3″]
Participants must be aged ≤ 6 weeks at first dose and can be of both genders. They must have a genetic 5q SMA homozygous deletion, mutation or compound heterozygous mutation; genetic documentation of 2 or 3 copies of survival motor neuron 2 (SMN2); compound muscle action potential (CMAP) ≥ 1 mV at Baseline; and gestational age of 37 to 42 weeks for singleton births or gestational age of 34 to 42 weeks for twins.
More information about the study can be found in the United States Clinical Trials Website at: www.clinicaltrials.gov (Clinical Identifier: NCT02386553).
Isis Pharmaceuticals also has two ongoing Phase 3 clinical studies that are examining the efficacy of their drug candidate. ENDEAR is a study assessing the efficacy of the drug in infants who have SMA type I and are already showing symptoms; and CHERISH is testing the use of ISIS-SMNRx in children ages 2-12 who have SMA type II.
The company also announced a fourth study called EMBRACE, a Phase 2 clinical study that will evaluate the efficacy and safety of their drug ISIS-SMNRx in patients with infantile or childhood-onset SMA. This study is expected to begin in the first half of 2015.